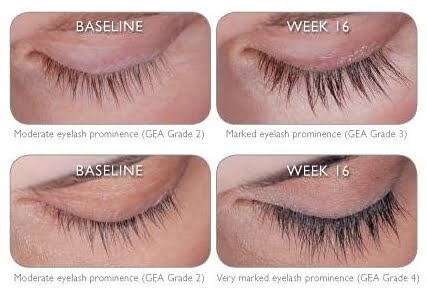
The new eyelash-enhancing drug Latisse is FDA approved and on the market. This hot new treatment was developed by Allergan, the company known for producing Botox and Juvéderm.
When it is applied to the base of the lashes, Latisse keeps hairs in their growth phase, producing longer,thicker and darker lashes. The effects are not permanent;the patient must keep using Latisse to maintain the look and eyelashes return to normal after discontinued use.
Latisse was developed almost by accident. Like many new drugs in recent years, positive side effects noticed during clinical trials prompted further research. In this case, a drug called Lumigan was being developed for glaucoma patients and during its third phase of trials, researchers noticed an interesting effect – significantly longer, fuller eyelashes. The active ingredient (bimatoprost) was reduced, and now Latisse is available for consumer use.
Call us at Boston Plastic Surgery for more details about Latisse!